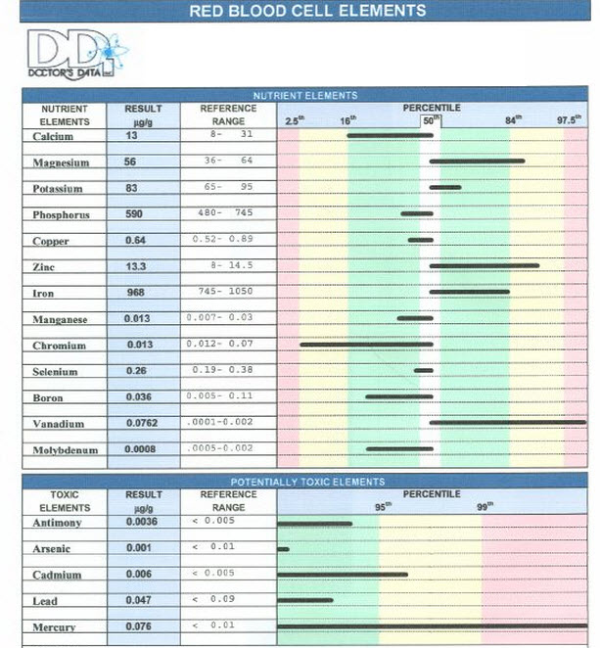
The Best Test for Mineral DeficienciesRonald Grisanti D.C., D.A.B.C.O., D.A.C.B.N., M.S.
The RBC Erthrocyte Blood Test can determine if you have mineral imbalances or...
By Gene Bruno, MS, MHS

In her 1962 groundbreaking book Silent Spring, Rachel Carson wrote: “For the first time in the history of the world every human being is now subjected to contact with dangerous chemicals, from the moment of conception until death.” Of course humans have always been exposed to potentially harmful chemicals from plants and other sources, but Rachel Carson’s point is well taken. Modern living exposes all of us to an unprecedented number of chemicals on a daily basis. This includes environmental toxins such as heavy metals, pesticides, industrial compounds and chemical byproducts, medications, cosmetic additives, inorganic chemicals, etc. These chemical substances which are foreign to the biological system are referred to as “xenobiotics.”
The good news is that the body was designed to detoxify and excrete xenobiotics. The bad news is our bodies may not always be equipped to handle the volume of modern, environmental pollutants and toxic substances. This problem may be exacerbated by the fact that the refining of many of our foodstuffs has caused them to provide considerably less of the nutrients that are essential to the detoxification process.1 2
The ramifications of toxic overload can vary from one individual to another. One possible ramification is multiple chemical sensitivities (MCS). MCS is a condition in which a person experiences various symptoms in response to being exposed to certain types of chemicals, primarily (but not limited to) those of petroleum and coal-tar derivation. The possible symptoms are many and may include headaches, fatigue, depression and an overall feeling of malaise and being sick. MCS seems to develop after consistent, long-term exposure to certain chemicals at home or in the workplace. Eventually, the person develops intolerance to these chemicals, and starts suffering from MCS. For many MCS people, a sensitivity reaction will occur when exposed to even minute amounts of the offending chemicals which, in turn, can lead to severe symptoms characteristic of the MCS condition.3
Of course an individual may suffer from toxic overload without having full-blown MCS. In fact, the most common symptom of toxic overload is probably fatigue. Other common symptoms include headache, muscle and joint pain, irritability, depression, mental confusion, gastrointestinal and/or cardiovascular irregularities, flu-like symptoms or allergic reactions including hives, stuffy or runny nose, sneezing and coughing.4 5
Furthermore, some researchers have suggested that toxic overload may contribute to autoimmune diseases including inflammatory and rheumatoid arthritis6 7, and neurological diseases such as Alzheimer's and Parkinson’s.8
The question of how to deal with toxic overload has a multipart answer which includes adapting to a healthier diet and reducing exposure to xenobiotics. The books Multiple Chemical Sensitivity by Gibson (2000, New Harbinger Publications, Inc.) and Staying Well in a Toxic World by Lawson (1993, The Nobel Press) provide good direction on how to do this. Another part to the answer is to help your liver to become more proficient at detoxification. That’s what this article is really about. So now let’s have a brief overview on how the liver goes about detoxification.
Water soluble toxins can pass through our bodies unchanged and be eliminated in the stool, sweat or urine. Fat soluble toxins, however, cannot be excreted without undergoing metabolic transformation (detoxification) in the liver so that they can become water soluble. Liver cells have sophisticated mechanisms to break down toxic substances. These include both endogenous (produced by the body) and exogenous (obtained from the environment; i.e., xenobiotics) substances. Every drug, chemical, pesticide and hormone, is broken down or metabolized via detoxification pathways in the liver called “phase 1” and “phase 2.” 9 10 11
Phase 1 utilizes cytochrome P450 enzymes produced in the liver. These enzymes initiate reactions that generally involve exposing or adding a “functional group” to the toxic molecule. This process of making the molecule more reactive is required as the first step in increasing its water solubility for excretion. Some chemicals are already highly reactive and they have functional groups, so they can bypass phase 1 and go right to phase 2. The majority, however, first need phase 1 activation. Unfortunately, phase 1 does generate free radicals which mean that there is greater potential for oxidative damage at this time.12 13
Phase 2 involves the coupling (attaching) or conjugation of a water soluble substance which is endogenously produced or sourced by the body, to the toxin. This makes the toxic molecule more water soluble and therefore less toxic. If the molecule is large, it is then excreted via the bile. Otherwise, it is excreted in the urine.14 15 There may even be a phase 3 for detoxification16, but that won’t be addressed in this article.
Now that we’ve briefly examined the process of liver detoxification, let’s take a look at how we can use dietary supplements to support and promote liver detoxification.
Certain amino acids are used in phase 2 as the water soluble substance that is conjugated (attached) to the toxic molecule. These amino acids include glycine, taurine, and glutamine.17 Clinically, supplementation with these amino acids has shown great benefit for patients with toxic overload, especially when body cleansing was undertaken contemporaneously.18 Likewise, methyl groups also act as conjugating agents in phase 2.19 Trimethylglycine (betaine) is a donor of methyl groups, and may be useful as such in biochemical reactions in the liver.20
In herbal medicine, Milk Thistle (Silybum marianum) is arguably the premium liver herb. The active component in Milk Thistle is its flavonoids collectively called silimarin; and the majority of Milk Thistle-related research has been conducted on this component. Silimarin has long been recognized for its ability to benefit people with liver disorders, including hepatitis21 22 23 24 and cirrhosis.25
Some toxic molecules pass through the glutathione conjugation pathway. A deficiency of this conjugating amino peptide can reduce the clearance of solvents from the bloodstream. Research shows that silimarin protects against glutathione depletion26, and increases liver glutathione status.27 Since glutathione is one of the primary conjugating agents in phase 2, this is a significant contribution by Milk Thistle in supporting detoxification by the liver.28
In addition, Milk Thistle also provides liver protection by stabilizing liver cell membranes. It alters the structure of the outer cell membrane in such a way as to prevent the penetration of the liver by toxins into interior of the cell. Milk Thistle also increases the regenerative ability of the liver and the formation of new liver cells. Further studies concluded that other actions of silimarin include preventing the recirculation of toxins and regeneration of damaged liver cells. Other studies indicate that Milk Thistle may prevent liver damage from liver poisoning prescription medications.29 30
Turmeric (Curcuma longa) is a bright yellow, ancient spice and a traditional remedy that has been used as a medicine, condiment and flavoring based on records dating back to 600 BCE. Turmeric and its curcuminoids also exhibit strong antioxidant activity31, enhance cellular resistance to oxidative damage32, and enhance the body's natural antioxidant glutathione levels; which in turn aids the liver in detoxification.33 Turmeric has also been found to have hepatoprotective (i.e., liver-protective) properties against a variety of liver-toxic chemicals and drugs.34 35 36 37 38
N-acetylcysteine (NAC) is the precursor to glutathione.39 It stimulates glutathione synthesis and promotes liver detoxification; as well as acting as a powerful scavenger of free radicals.40 41 Historically the most prevalent and well- accepted use of NAC has been as an antidote for acetaminophen (Tylenol®, paracetamol) poisoning.42 The resultant liver toxicity is due to an acetaminophen metabolite that depletes the liver cells of glutathione and causes liver cell damage and possibly even death. NAC has also been effective for heavy metal poisoning by gold, silver, copper, mercury, lead, and arsenic, as well as in cases of poisoning by carbon tetrachloride, acrylonitriles, halothane, paraquat, acetaldehyde, coumarin, and interferon.43 Since detoxification of mercury depletes glutathione, the use of NAC doubly makes sense.

Green tea (Camellia sinensis) compounds increase both phase 1 and phase 2 enzyme activities.44 Research shows that green tea may have liver protective properties.45 46 47 48 In addition, green tea has been shown to block chromosomal (DNA) damage from chemicals in cigarette smoke.49 Perhaps not surprisingly, green tea polyphenols are associated with a reduced risk of certain cancers in humans.50 Other research has also shown an anticancer effect from these polyphenols51, including an inhibition of metastasis in skin cancer cells.52
Another phase 2 pathway is the glucuronidation pathway. In this pathway, glucuronic acid is attached to certain toxins as well as hormones such as estrogen to facilitate their removal by excreting them via bile into the intestinal tract.
The problem is that beta-glucuronidase, a bacterial enzyme found in the intestines, can break the bond that attaches the glucuronic acid to the toxin. Now the toxin can be reabsorbed back into the bloodstream, thereby contributing to the total toxic load on the liver; not a good thing. There is, however, a way to inhibit beta- glucuronidase from breaking the bond in the first instance. It involves D-glucarate, or calcium D- glucarate.
D-glucaric acid is a natural substance found in many fruits and vegetables such as apples, grapefruit, broccoli, and Brussels sprouts. Calcium D-glucarate is the calcium salt of D- glucaric acid typically used in dietary supplements. Calcium D-glucarate has been shown to inhibit beta-glucuronidase.53 As a matter of fact according to data released from the University of Texas M.D. Anderson Cancer Center, Calcium D-glucarate inhibited beta- glucuronidase by 57% in the blood, 44% in the liver, 39% in the intestines, and 37% in the lungs.54 Such an inhibition of beta- glucuronidase may do much to protect the action of the glucuronidation pathway.
In traditional Chinese medicine, Schizandra (Schizandra chinensis) is mainly used to treat cough and wheezing, spontaneous sweating, nocturnal emission, chronic diarrhea, insomnia and forgetfulness.55 In Russia, it is regarded as an adaptogen.56 Research indicates that Schizandra can improve work performance, build strength, reduce fatigue, and increase endurance57 58; all consistent with the effects of an adaptogen.
Schizandra also appears to have significant value for the liver. For example, it appears that the lignans in Schizandra protect the liver by activating the enzymes in liver cells that produce the antioxidant glutathione,59 so this herb is beneficial for phase 2 detoxification reactions. Furthermore, in-vitro (i.e., test tube) studies have shown that constituents of Schizandra decrease the mutagenicity of mutagenic toxin benzo(a)pyrene (BaP).60 Schizandra also induces phase 1 detoxification enzymes61, yet has not been shown to cause any drug interactions. Schizandra’s Studies from China indicate that Schizandra helped patients with chronic viral hepatitis.62 63
Garlic appears to induce both phase 1 and phase 2 enzymes.64 65 In addition, aged garlic extract may increase the glutathione level and glutathione related enzymes which aid in detoxifying the body.66 67 68 69 Research has also suggested that garlic extract may help to protect the body from heavy metal poisoning.70 When garlic extract was combined with red blood cells it prevented lead, mercury and aluminum from destroying them. Without the garlic extract, these heavy metals ruptured the red blood cells.
Rosemary contains carnosol, an antioxidant that inhibits bioactivation of BaP and induces glutathione-S-transferase (a detoxification enzyme) and other important phase 2 enzymes.71 72 In a study73 where mice were given Rosemary, the researchers concluded that liver activities of phase 2 liver enzymes were significantly increased, and that the results indicated that components of rosemary extract have the potential to protect the liver and stomach from carcinogenic or toxic agents.
Bladderwrack (Fucus vesiculosus) is a type of brown algae that grows on the northern coasts of the Atlantic, Pacific and Baltic. In research74, Bladderwrack has been shown to consume and metabolize toxic polycyclic aromatic hydrocarbons, particularly BaP. Likewise, Bladderwrack has been shown to absorb certain heavy metals such as tin.75 Finally, an antibacterial compound has been isolated from Bladderwrack, and is effective against various Gram-positive and Gram-negative bacteria.
The dietary supplements discussed in this article are certainly not the only natural compounds capable of supporting and facilitating the liver’s detoxification process. Compounds from citrus fruit and the Brassica or cruciferous group of vegetables (e.g., cabbage, Brussels sprouts, broccoli, etc.), as well as the dietary supplement alpha lipoic acid are some examples of other effective detoxifying agents.76 Nevertheless, the compounds listed can be effective adjuncts to a program for detoxification.
Although the focus of this article is on the use of dietary supplements which may help promote liver detoxification, it should be understood that toxic overload should be treated with a comprehensive approach that also includes adapting to a healthier diet and reducing exposure to xenobiotics. Furthermore, since there can be side effects associated with a serious program of detoxification, it should generally be attempted with the help of a healthcare professional who is knowledgeable about the process.
Smart SupplementationTM is a free series of educational literature created by Huntington College of Health Sciences (HCHS) as a public service. Although copyrighted, it may be freely photocopied and distributed, but may not be altered in any way. Smart SupplementationTM is not intended as medical advice. For diagnosis and treatment of any medical condition, consult your physician.
1. Rogers SA. Chemical Sensitivity: Breaking the Paralyzing Paradigm. Internal Medicine World Report 1992; February 1-14:15-16. 2. Rogers SA. Chemical Sensitivity: Breaking the Paralyzing Paradigm. Internal Medicine World Report 1992; March 15-31: 8-31. 3. Lawson L. Staying Well in a Toxic World. Chicago: The Nobel Press, Inc.; 1993. 4. Lawson L. Staying Well in a Toxic World. Chicago: The Nobel Press, Inc.; 1993. 5. Gibson PR. Multiple Chemical Sensitivity. Oakland, California: New Harbinger Publications, Inc.; 2000. 6. Rooney PJ, Jenkins RT, Buchanan WW. A short review of the relationship between intestinal permeability and inflammatory joint disease. Clinical and Experimental Rheumatology 1990; 8(1):75-83.7. Smith MD, Gibson RA, Brooks PM. Abnormal bowel permeability in ankylosing spondylitis and rheumatoid arthritis. Journal of Rheumatology 1985; 12(2):299-305. 8. Steventon, GB, Heafield MT, Sturman S, Waring RH, Williams AC. Xenobiotic metabolism in Alzheimer's disease. Neurology, 1990;40(7):1095-1098. 9. Murray Rk, Granner DK, Mayes PA, Rodwell VW. Harper’s Biochemistry, 25th ed. New York: McGraw Hill; 200:780-786. 10. Lüllmann H. Mohr K, Ziegler A, Bieger D. Color Atlas of Pharmacology, 2nd ed. Stuttgart: Thieme; 2000:32-39. 11. Roundtree R. The Use of Phytochemicals in the Biotransformation and Elimination of Environmental Toxins. IN Medicines from the Earth 2003: Official Proceedings. Brevard, North Carolina: Gaia Herbal Research Institute; 2003:115-128. 12. Murray Rk, Granner DK, Mayes PA, Rodwell VW. Harper’s Biochemistry, 25th ed. New York: McGraw Hill; 200:780-786. 13. Lüllmann H. Mohr K, Ziegler A, Bieger D. Color Atlas of Pharmacology, 2nd ed. Stuttgart: Thieme; 2000:32-39. 14 Murray Rk, Granner DK, Mayes PA, Rodwell VW. Harper’s Biochemistry, 25th ed. New York: McGraw Hill; 200:780-786. 15. Lüllmann H. Mohr K, Ziegler A, Bieger D. Color Atlas of Pharmacology, 2nd ed. Stuttgart: Thieme; 2000:32-39. 16. Liska DJ. The Detoxification Enzyme Systems. Alternative Medicine Review 1998;3(3):187-198. 17. Crinnion WJ. Environmental Medicine, Part 2 – Health Effects of and Protection from Ubiquitous Airborne Solvent Exposure. Alternative Medicine Review 2000; 5(2):133-143 18. Hill RH Jr, Ashley DL, Head SL, et al. p- Dichlorobenzene exposure among 1,000 adults in the United States. Arch Environ Health 1995;50:277-280. 19. Roundtree R. The Use of Phytochemicals in the Biotransformation and Elimination of Environmental Toxins. IN Medicines from the Earth 2003: Official Proceedings. Brevard, North Carolina: Gaia Herbal Research Institute; 2003:115-128. 20.Anonymous. Betaine: Monograph. Alternative Medicine Review 2003; 8(2):193-196. 21. Vailati A, Aristia L, Sozze E, et al. Randomized open study of the dose-affect relationship of a short course of IdB 1016 in patients with viral or alcoholic hepatitis. Fitoterapia 1993; 64:219-27. 22.Lirussi F, Okolicsanyi L. Cytoprotection in the nineties: experience with ursodeoxycholic acid and silymarin in chronic liver disease. Acta Physiol Hung 1992; 80:363-7 23. Magliulo E, Gagliardi B, Fiori GP. Results of a double blind study on the effect of silymarin in the treatment of acute viral hepatitis carried out at two medical centers. Med Klin 1978; 73:1060-5. 24. Bode JC, Schmidt U, Durr HK. Silymarin for the treatment of acute viral hepatitis? Report of a controlled trial. Med Klin 1977; 72:513-8. 25. Schuppan D, Strösser W, Burkard G, Walosek G. Legalon® lessens fibrosing activity in patients with chronic liver diseases. Zeits Allgemeinmed 1998; 74:577-84. 26. Campos R, Garido A, Guerra R, et al. Silybin dihemisuccinate protects against glutathione depletion and lipid peroxidation induced by acetaminophen on rat liver. Planta Med 1989;55:417-419. 27. Valenzuela A, Aspillaga M, Vial S, Guerra R. Selectivity of silymarin on the increase of the glutathione content in different tissues of the rat. Planta Med 1989; 55(5):420-2. 28 Crinnion WJ. Environmental Medicine, Part 2 – Health Effects of and Protection from Ubiquitous Airborne Solvent Exposure. Alternative Medicine Review 2000; 5(2):133-143 29. Blumenthal M. Herbal Medicine, Expanded Commission E Monographs, 1st ed. Austin: American Botanical Council; 2000. 30. Brinker F. Herb Contraindications and Drug Interactions, 2nd ed. Sandy (OR): Eclectic Med; 1998. 31. Toda S, Miyase T, Arich H, et al. Natural antioxidants. Antioxidative compounds isolated from rhizome of Curcuma longa L. Chem Pharmacol Bull 1985;33:1725- 1728. 32. Mortellini R, Foresti R, Bassi R, Green CJ. Curcumin, an antioxidant and anti-inflammatory agent, induces heme oxygenase-1 and protects endothelial cells against oxidative stress. Free Radic Biol Med 2000;28:1303-1312. 33. Pizorrno JE, Murray MT. Textbook of Natural Medicine, 2nd Ed. London: Churchill Livingstone; 1999;689-693. 34. Deshpande UR, Gadre SG, Raste AS, et al. Protective effect of turmeric (Curcuma longa L.) extract on carbon tetrachloride-induced liver damage in rats. Indian J Exp Biol 1998;36:573-577. 35. Park E J, Jeon CH, Ko G, et al. Protective effect of curcumin in rat liver injury induced by carbon tetrachloride. J Pharm Pharmacol 2000;52:437-440. 36. Kiso Y, Suzuki Y, Watanabe N, et al. Antihepatotoxic principles of Curcuma longa rhizomes. Planta Med 1983;49:185-187. 37. Donatus IA, Sardjoko, Vermeulen NP. Cytotoxic and cytoprotective activities of curcumin. Effects on paracetamol-induced cytotoxicity, lipid peroxidation and glutathione depletion in rat hepatocytes. Biochem Pharmacol 1990;39:1869-1875. 38. Soni KB, Rajan A, Kuttan R. Reversal of aflatoxin induced liver damage by turmeric and curcumin. Cancer Lett 1992;66:115-121. 39. Anonymous. Alternative Medicine Review 2000; 5(5); 467-471. 40. De Vries N, De Flora; S. N-Acetyl-l-Cysteine. J Cell Biochem 1993;17F:S270-S277.(- 41. De Flora S, Bennicelli C, Camoirano A, et al. In vivo effects of N-acetylcysteine on glutathione metabolism and- on the biotransformation of carcinogenic and/or mutagenic compounds. Carcinogenesis 1985;6:1735-1745. 42. Anonymous. Alternative Medicine Review 2000; 5(5); 467-471. 43. Zimet I. Acetylcysteine: A drug that is much more than a mucokinetic. Biomed & Pharmacother 1988;42:513-520. 44. Stoner GD, Mukhtar H. Polyphenols as cancer chemopreventive agents. J Cell Biochem Suppl 1995; 22:169-80. 45. Presser A. Pharmacist’s Guide to Medicinal Herbs. Petaluma, California:Smart Publications; 2000. 46. Kuhn MA, Winston D. Herbal Therapy & Supplements: A Scientific & Traditional Approach. Philadelphia:Lippincott; 2000. 47. Mills S, Bone K. Principles and Practice of Phytotherapy: Edinburgh;Churchill Livingstone; 2000. 48. Blumenthal M, Goldberg A, Brinckmann J (eds). Herbal Medicine: Expanded Commission E Monographs: Newton, Massachusetts;Integrative Medicine Communications; 2000. 49. Shim JS, Kang MH, Kim YH, Roh JK, Roberts C, Lee IP. Chemopreventive effect of green tea (Camellia sinensis) amonth cigarette smokers. Cancer Epidemiol Biomakers Prev 1995; 4(4):387-391. 50. Mukhtar H, Ahmad N. Green tea in chemoprevention of cancer. Toxicol Sci 1999;52(2 Suppl):111–7. 51. Suganuma M, Okabe S, Sueoka N, et al. Green tea and cancer chemoprevention. Mutat Res 1999;428:339–44. 52. Menon LG, Kuttan R, Kuttan G. Anti-metastatic activity of curcumin and catechin. Cancer Lett 1999;141:159–65. 53. Walaszek Z, Szemraj J, Narog M. Metabolism, uptake, and excretion of a D-glucaric acid salt and its potential use in cancer prevention. Cancer Detect Prev 1997; 21:178-90. 54. Dwivedi C, Heck WJ, Downie AA, et al. Effect of calcium glucarate on beta-glucuronidase activity and glucarate content of certain vegetables and fruits. Biochem Med Metab Biol 1990;43:83–92. 55. Chang HM, But PP (eds). Pharmacology and Applications of Chinese Materia Medica, Vol 1. Singapore: World Scientific; 1986. 56. Bone K. Clinical Applications of Ayurvedic and Chinese Herbs. Warwick, Queensland:Phytotherapy Press; 1996:69- 74. 57. Foster S, Yue CX. Herbal Emissaries: Bringing Chinese Herbs to the West. Rochester, VT: Healing Arts Press; 1992:146–52. 58. Fulder S. The Root of Being. London: Hutchinson and Co; 1980. 59. Ip SP, Poon MKT, Wu SS, et al. Effect of schisandrin B on hepatic glutathione antioxidant system in mice: Protection against carbon tetrachloride toxicity. Planta Med 1995; 61:398-401. 60. Bone K. Influence of Herbs on Detoxification by the Liver.. IN Medicines from the Earth 2003: Official Proceedings. Brevard, North Carolina: Gaia Herbal Research Institute; 2003:1-11. 61. Zhu M, et al. Evaluation of the protective effects of schisandra chinensis on phase I drug metabolism using a CCl4 intoxication model. J Ethnopharmacol 1999; 67:61-8. 62. Liu KT. Studies on fructus Schizandrae chinensis. Annex 12: Studies on fructus Schizandrae chinensis. Plenary lecture, World Health Organization (WHO) Seminar on the Use of Medicinal Plants in Health Care, Sept 1977, Tokyo, Japan. In: WHO Regional Office for the Western Pacific, Final Report, November 1977, Manila, 101–12. 63. Chang HM, But PP (eds). Pharmacology and Applications of Chinese Materia Medica, Vol 1. Singapore: World Scientific; 1986. 64. Manson MM, Ball HW, Barrett MC, et al. Mechanism of action of dietary chemoprotective agents in rat liver: induction of phase I and II drug metabolizing enzymes and aflatoxin B1 metabolism. Carcinogenesis 1997; 18(9):1729-38. 65. Dalvi RR. Alterations in hepatic phase I and Phase II biotransformation enzymes by garlic oil in rates. Toxicol Lett 1992; 13(10): 1921-23. 66. Li G, Qiao CH, Lin RI, Pinto JT, Osborne MP, Tiwari RK. Antiproliferative effects of garlic constituents on cultured human breast cancer cells. Oncol. Rep 1995; 2:787-791. 67. Tadi PP, Teel RW, Lau BHS. Organosulfur compounds of garlic modulate mutagenesis, metabolism, and DNA binding of aflatoxin B1. Nutr. Cancer 1991; 15:87-95 68. Sumiyoshi H, Wargovich MJ. Chemoprevention of 1,2- dimethylhydrazine-induced colon cancer in mice by naturally occurring organosulfur compounds. Cancer Res 1990; 50:5084-5087 69. Wargovich, M. J. (1987) Diallyl sulfide, a flavor component of garlic (Allium sativum), inhibits dimethylhydrazine-induced colon cancer. Carcinogenesis 8:487-489. 70. Lau BH. 1989. Detoxifying Radioprotective and Phagocyte-enhancing Effects of Garlic. Int. Clin. Nutr. Rev. 9(1): 27-31. 71. Singletary KW. Rosemary extract and carnosol stimulate rat liver glutathione-S-transterase and quinine reductase activities. Cancer Lett 100(1-2):139-144. 72. Offord EA, Mace K, Ruffieux C, et al. Rosemary components inhibt benzo{a}pyrene-induced genotoxicity in human bronchial cells. Carcinogenesis 1995; 16(9):2057- 2062. 73. Singletary KW, Rokusek JT. Tissue-specific enhancement of xenobiotic detoxification enzymes inmice by dietary rosemary extract. Plant Foods Hum Nutr. 1997;50(1):47-53. 74. Kirso U, Irha N. Role of algae in fate of carcinogenic polycyclic aromatic hydrocarbons in the aquatic environment. Ecotoxicology and environmental safety 1998; 41:83-9. 75. Wright PJ, Weber JH. Biosorption of inorganic tin and methyltin compounds by estuarine macroalgae. Environmental Science and Technology 1991; 25(2)287- 294. 76. Roundtree R. The Use of Phytochemicals in the Biotransformation and Elimination of Environmental Toxins. IN Medicines from the Earth 2003: Official Proceedings. Brevard, North Carolina: Gaia Herbal Research Institute; 2003:115-128.

The Best Test for Mineral DeficienciesRonald Grisanti D.C., D.A.B.C.O., D.A.C.B.N., M.S.
The RBC Erthrocyte Blood Test can determine if you have mineral imbalances or...
Ronald Grisanti D.C., D.A.B.C.O., D.A.C.B.N., M.S.
Just recently I had the opportunity to consult with a patient by the name of...